Cell Culture Media
Overview
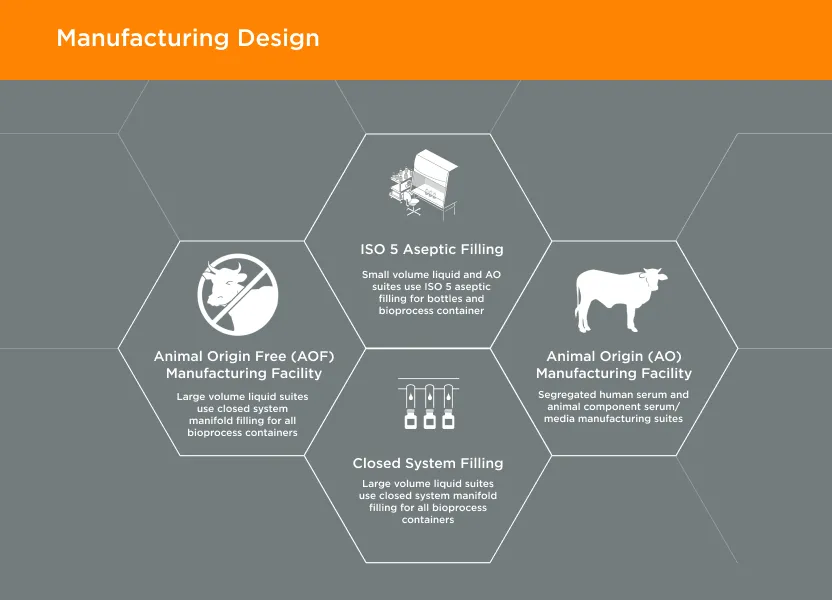
GeminiBio offers a wide range of specialized cell culture media options, including insect cell media, neuronal media, T-Cell media, as well as other media for specific applications. In addition to our standard “off the shelf” cell culture media products, GeminiBio can provide custom manufacturing to meet customer’s unique media specifications. Our cell culture media is manufactured under cGMP to meet customers quality requirements and improve their workflow productivity, reproducibility, and speed.